Amperometric sensors work by taking an electrical signal and translating it to a concentration. The graphic below illustrates this process.

First, the analyte of interest (chlorine, chlorine dioxide, ozone, etc.) gets reduced on the measuring electrode of the sensor. This generates a raw electrical signal, measured in millivolts (mV). This signal gets processed by the Neon® analyzer, which uses your pH, calibration slope, temperature, and other parameters to turn the mV signal into a concentration value, usually measured in ppm or ppb. This concentration is what gets displayed on the Neon® unit.
In order for the analyzer to make the conversion between an electrical signal to a concentration, a calibration must be performed. A calibration tells the analyzer "an electrical signal of ___ mV corresponds to a disinfectant concentration of ___ ppm" where the disinfectant concentration is measured with a reference method such as DPD. This process is shown below.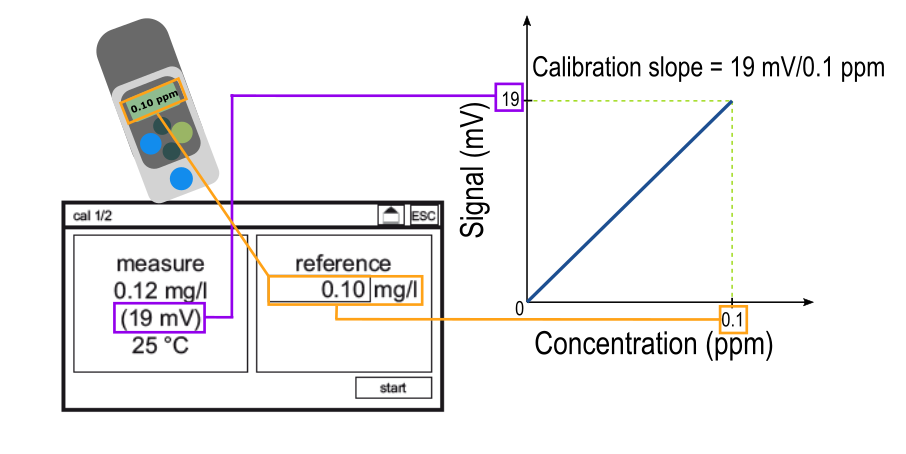
Inputting a reference value on the calibration screen generates a calibration slope. For each measuring range, there is an "ideal" slope, which you can learn more about here. The calibration slope is the tool that the analyzer uses to translate between an electrical signal and a concentration. Thus, the quality of the calibration determines the quality of the measurements made after the calibration is performed. The graphic below depicts the difference between a "good" and a "bad" calibration.
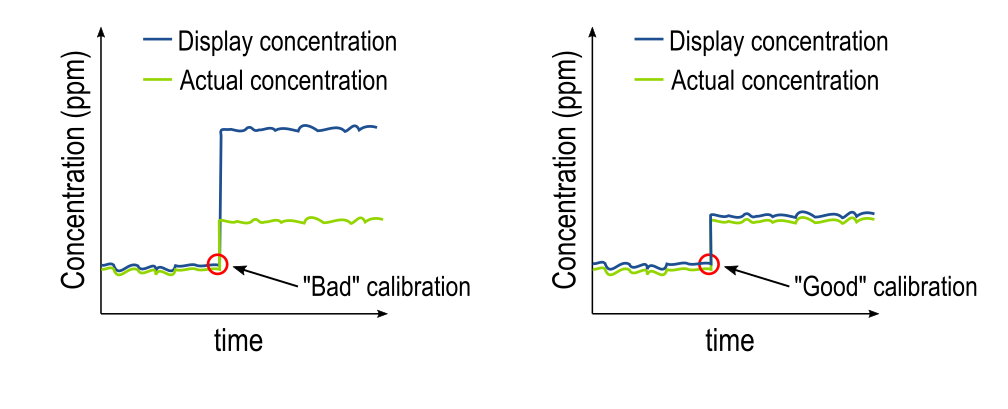
The plot on the left shows the effect of a "bad" calibration, where the reference value input was much higher than the actual disinfectant concentration, which caused a false high reading on the display. The false high reading from the reference measurement could have been caused by many factors, such as sampling errors, chemical interferences, or physical interferences, which are all common when using DPD as a reference. You can learn more about the limitations of DPD as a reference measurement here.
The plot on the right shows the effect of a "good" calibration, where the reference measurement matched well with the actual disinfectant concentration. To achieve a good calibration like the plot on the right, try taking three reference measurements back-to-back and average them together. Always be sure to follow the instructions of your reference measurement system to get the best results.
Calibration is very important to get accurate concentration readings. If you have any questions about calibration or how it works, contact a Kuntze representative or submit a ticket.
